As a part of their Digital Transformation initiatives, increasing regulatory scrutiny and requirements to comply with national and international regulations, companies are rapidly replacing manual and paper-based processes with electronic systems. Due to their experience and background, CIMCON Software consultants are well equipped to qualify your digital systems of varying levels of complexity and help you stay compliant. CIMCON optimizes the time and cost of validation by applying a risk-based approach to each system. CIMCON can also provide a wide range of automated tools as part of the consulting engagement to provide you with sustainable compliance.
CIMCON is a recognized leader in providing compliance solutions to the FDA-regulated industry with a global client base that spans North America, Europe and Asia. CIMCON consultants combine regulatory knowledge with technical expertise to provide solutions to issues instead of simply highlighting them. CIMCON’s validation methodologies have been developed after years of real-world field experience to ensure successful project outcomes, and include widely followed practices such as GAMP 5. We strive to deliver value as an added ingredient in all our deliverables with a view to improving operations and productivity and simplifying processes with compliance as a by-product.
CIMCON offers a wide range of services that include validation planning, risk assessments, Part 11 assessments and remediation plans, preparation of validation documents and SOPs, protocol execution, deviation resolution and training. CIMCON regularly contributes to industry conferences, journals, tradeshows and events and is in tune with current FDA thinking and future direction.
Representative Projects
CIMCON’s consulting services group has extensive experience in providing 21 CFR Part 11 Compliance and Validation Services that include the following:
21 CFR Part 11 Services
- Gap Assessment
- Strategy and Planning
- System Inventory
- Remediation
- Software Development
- Technical Controls
- Procedural Controls
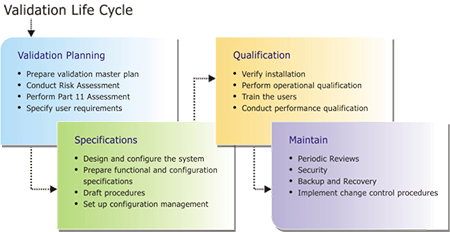
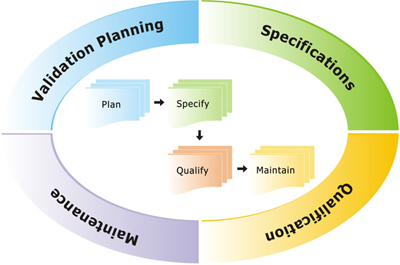
Planning
- Master Validation Plan(MVP)
- Risk Assessment
Requirements
- User Requirements Specification (URS)
Design
- Software Design Specification (SDS)
- Hardware Design Specification (HDS)
Configuration
- Hardware Configuration Specification (HCS)
- Application Configuration Specification (ACS)
Specifications
- Functional Specifications (FS)
- Standard Operating Procedures (SOPs)
Testing
- Installation Qualification (IQ)
- Operational Qualification (OQ)
- Performance Qualification (PQ)
Reporting
- Summary Reports
Computer Systems Validation and Part 11 Assessment Experience
– Representative Systems
- Lab Equipment/Instrument Software
- Medical Devices
- Enterprise Resource Planning (ERP) Systems
- Laboratory Information Management System (LIMS)
- Quality Management Systems (QMS)
- Adverse Event Systems
- CAPA Systems
- Network Infrastructure Qualification
- Laboratory Systems
- Spreadsheet Validation
- PLC, DCS and Plant Floor Systems
- Equipment/Systems Validation
- Label Management Systems
- Manufacturing Execution Systems (MES)
- Clinical Trial Management Systems
- Source Code Reviews
- Change Management Systems
- Batch/Process Data Management System
- Database Querying and Information Retrieval
- Computerized Maintenance Management Systems
- Environmental Monitoring and Trending
- Training Systems
- HPLC Systems

